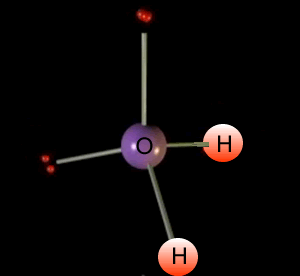
As the lone pairs take up more space, the bond angle is less than the ideal 109.5 VSEPR bond angle. 
The H-O bond length is 0.958 Å (1 Å 10 10 m). Molecular orbital theory (MO theory) provides an explanation of chemical bonding that accounts for the paramagnetism of the oxygen molecule. This is consistent with a roughly tetrahedral orientation of four electron domains about the central oxygen, two bonding pairs and two non-bonding 'lone pairs'. The atomic orbitals (also called basis functions) are labeled as s, for example, 1s and 3pz or simply as 1 and 2. In agreement with our analysis using the Lewis formalism, waters shape is angular, or bent, with an H-O-H bond angle of 104.5°. Molecular orbitals are also called wavefunctions (), because they are solutions to the Schrödinger equation for the molecule. A water molecule contains two hydrogen atoms, each of which share an electron with oxygen forming covalent bonds. 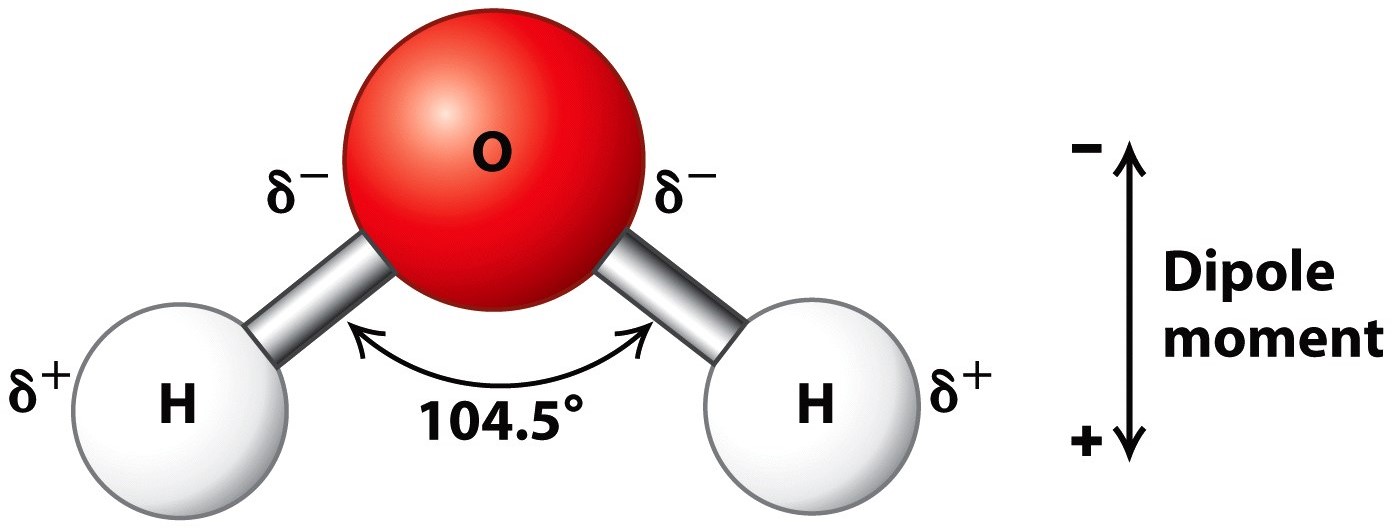
The four electron groups are the 2 single bonds to Hydrogen and the 2 lone pairs of Oxygen. In addition one can see that the electron density is delocalized over the whole molecule. \): Electron configuration of water with oxygen's 6 electrons (black) in four sp 3 hybridized orbitals, and 2 \(\sigma\) bonds arising from the two orbitals that only have one electron from oxygen (black) and one from hydrogen (red). 1: Sigma bonding and antibonding combinations of an s and p orbital. Water has four electron groups so it falls under tetrahedral for the electron-group geometry. The structure is V-shaped with a bond angle of 103.7.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
